Atomic Theory: Electrons and Holes
A beginner-friendly exploration of how electrons and holes behave inside atoms and materials—and how this invisible physics powers modern technology from smartphones to solar cells.
Table of Contents
- Introduction: Why Electrons and Holes Matter
- Atomic Structure Basics
- How Electrons Move Through Materials
- What Exactly Is a Hole?
- Semiconductor Behavior: The Electron–Hole System
- Technology Applications of Electrons and Holes
- Top 5 Frequently Asked Questions
- Final Thoughts
- Resources
Introduction: Why Electrons and Holes Matter
Electrons and holes might sound like abstract physics concepts, but they are central to everything in our digital world. Every smartphone display, computer processor, LED light, and solar panel depends on how these tiny particles and “missing particles” behave inside atoms and materials.
Understanding electrons and holes reveals the invisible engine powering technological innovation. Even at a beginner level, grasping these fundamentals unlocks an understanding of semiconductors—the backbone of modern electronics.
This article simplifies atomic theory, explores how electrons move, and explains why holes—despite being “missing electrons”—play an essential role in making technology work.
Atomic Structure Basics
Atoms and Their Key Components
Every material in the universe is made of atoms, which consist of three main parts:
- Protons (positive charge)
- Neutrons (neutral charge)
- Electrons (negative charge)
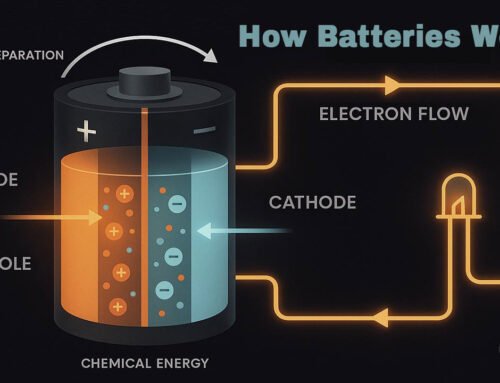
Electrons orbit the nucleus and define how an atom interacts with electricity. Their motion and energy determine whether a material can conduct a current.
How Electrons Store and Use Energy
Electrons do not orbit randomly; they exist in organized layers called **energy levels** or **shells**.
The important ideas are:
- Electrons closer to the nucleus have lower energy.
- Electrons farther away have higher energy.
- The outermost electrons, called **valence electrons**, determine conductivity.
When energy is applied (heat, light, voltage), electrons can jump between energy levels. This movement is the foundation for electrical conduction.
How Electrons Move Through Materials
Conductors, Insulators, and Semiconductors
Materials behave differently depending on how tightly they hold their electrons.
- Conductors (like copper):
Electrons move freely. Great for electrical wiring. - Insulators (like rubber):
Electrons are tightly bound. They block current. - Semiconductors (like silicon):
Electrons move sometimes. Their conductivity can be engineered.
This tunable property makes semiconductors the perfect foundation for electronics.
Understanding Band Theory
To explain conduction, scientists use **band theory**:
- Valence band: where electrons usually reside
- Conduction band: where electrons can move freely to conduct electricity
- Band gap: the energy difference between the two
A small band gap allows electrons to jump into the conduction band. This is exactly how semiconductors operate—and why they enable precise electronic control.
What Exactly Is a Hole?
A **hole** is simply a missing electron in the valence band. But this absence behaves like a positively charged particle.
When an electron moves to fill the empty space, the hole appears to shift in the opposite direction.
This creates the illusion that holes “move,” even though they are literally nothing more than vacant positions.
In semiconductor physics, holes are treated as real particles because their behavior influences current flow.
How Holes Move (Even Though They Don’t Exist)
Imagine a row of seats with one empty chair.
When people shift over to fill the seat, the empty space appears to move backward.
This is exactly how holes move.
The movement of the hole is simply the chain reaction of electrons shifting to fill the gap.
Why Holes Are as Important as Electrons
Electrons carry negative charge; holes behave like positive charge carriers.
In semiconductors, **both are necessary** for controlling current.
Modern electronics rely on the careful choreography of electrons and holes working together.
Semiconductor Behavior: The Electron–Hole System
Doping: Engineering Electron and Hole Concentrations
Pure silicon is not very conductive.
Engineers “dope” silicon by adding tiny amounts of other elements to control electrons and holes.
Two types of doping:
- N-type: adds electrons
- P-type: creates holes
This engineered balance allows electronics to function with incredible precision.
How PN Junctions Control Charge
A **PN junction** forms when P-type and N-type materials are joined.
This junction acts like a gate:
- It allows current to flow one way.
- It blocks current from flowing the other way.
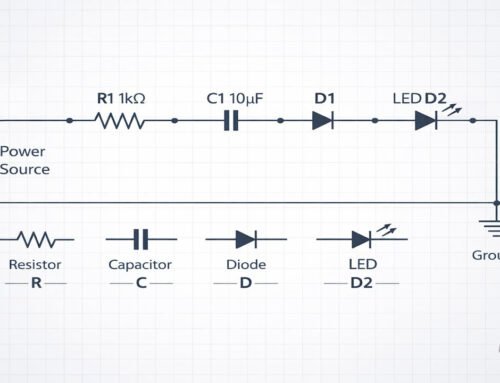
Diodes, LEDs, and transistors all rely on PN junctions.
Technology Applications of Electrons and Holes
Transistors and Digital Logic
Transistors are the building blocks of chips.
They use electrons and holes to switch signals on and off.
More than 100 billion transistors can fit inside a modern smartphone processor.
Solar Cells and Energy Conversion
Solar cells work by absorbing sunlight, which excites electrons into the conduction band.
This creates electron–hole pairs that generate electricity.
LEDs and Light Emission
In LEDs, electrons and holes recombine.
This recombination releases energy as light—a process known as **electroluminescence**.
Different materials produce different colors.
Top 5 Frequently Asked Questions
Final Thoughts
The most important takeaway is that electrons and holes are the fundamental drivers of modern electronics.
Electrons provide mobile negative charge, while holes act as positive charge carriers created when electrons move. Together, they form the dynamic system that makes semiconductors—and thus all digital technology—possible.
Understanding this invisible interplay gives beginners a solid foundation in how devices from phones to solar panels work at the atomic level.






Leave A Comment